egnite, Inc. and JenaValve Announce Strategic Partnership to Uncover Trends in Care for Patients with Aortic Regurgitation
Partnership aims to understand the care paradigm and associated outcomes for patients with aortic regurgitation
First study presented by Nick Amoroso, MD, at the TVT Structural Heart Summit in Phoenix, Arizona
ALISO VIEJO, Calif. – June 9, 2023 – egnite, Inc., a leading digital health company for cardiovascular care, and JenaValve Technology, Inc., a developer and manufacturer of differentiated transcatheter aortic valve replacement (TAVR) systems, are pleased to announce a strategic partnership aimed to shed light on the care paradigm and associated outcomes of patients with aortic regurgitation (AR). The partnership leverages egnite’s industry-leading database to quantify prevalence and treatment patterns and has conservatively estimated there may be over 500,000 patients in the United States diagnosed with moderate or severe AR.
Duane Pinto, MD, CMO of JenaValve, further emphasized the need for broader access to therapies, stating, “By utilizing cutting-edge analytics and JenaValve’s innovative transcatheter therapies, we hope to drive urgent change, provide improved treatment options, and ultimately save lives. These findings also provide valuable insights for healthcare providers and policymakers, encouraging further attention toward improving outcomes for patients suffering from AR.”
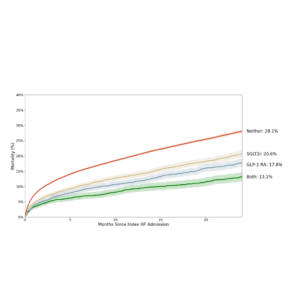
Interventional cardiologist Dr. Nicholas S. Amoroso presented the first study from this partnership at TVT: The Structural Heart Summit. This novel study examined two-year treatment rates and outcomes for patients with AR utilizing over 1 million patients from 25 leading teaching and non-teaching cardiovascular programs across the United States from egnite’s database.
Key findings uncovered that despite high two-year mortality rates and guideline indications for treatment, patients with severe AR received low rates of specialist evaluations and treatment with aortic valve replacement. The study also demonstrated that patients diagnosed with moderate or severe AR and severe left ventricular dilation exhibited similar mortality rates, suggesting that some patients may benefit from earlier assessment by the Heart Team.
“This is one of the first artificial intelligence-driven studies providing critical insights into care patterns for patients with moderate or greater AR in the community,” stated Nicholas S. Amoroso, Assistant Professor at the Medical University of South Carolina. “The urgency for digital technologies to identify AR patients earlier and novel therapies to improve outcomes for this vulnerable patient population has never been greater.”
About JenaValve
JenaValve Technology, Inc. is a medical device company focused on the design, development, and commercialization of innovative transcatheter heart valve solutions for the treatment of patients suffering from heart valve disease. The Company’s Trilogy® Heart Valve System is a transcatheter aortic valve replacement (TAVR) system designed to treat patients with symptomatic, severe aortic regurgitation (ssAR) and symptomatic, severe aortic stenosis (ssAS) in high surgical risk patients. The Trilogy System received CE Mark approval in May 2021, providing European physicians with the first TAVR device with true dual-disease treatment capabilities.
JenaValve received Breakthrough Device Designation from the FDA to facilitate the review of ALIGN-AR, a pivotal trial in the U.S., as part of its PMA application for the Trilogy Heart Valve System for the treatment of ssAR in high surgical risk patients. If approved, the Trilogy System would become the first and only TAVR system in the U.S. indicated for the treatment of AR.
JenaValve is headquartered in Irvine, California, with additional locations in Leeds, U.K., and Munich, Germany.
JenaValve is backed by Bain Capital Life Sciences, Andera Partners (formerly Edmond de Rothschild Investment Partners), Valiance Life Sciences, Rosetta Capital, Cormorant Asset Management, Legend Capital, NeoMed Management, RMM, VI Partners, Pictet Alternative Advisors SA, Qatar Investment Authority (QIA), Innovatus Capital Partners, and Peijia Medical Limited.
US: CAUTION – Investigational device. Limited by Federal (or United States) law to investigational use.
Additional information is available at www.jenavalve.com
About egnite, Inc.
egnite is a visionary digital health company committed to advancing the health of our society through innovative cardiovascular solutions. egnite uses AI-driven algorithms and big data to produce business intelligence for healthcare, elevating the role of data in critical decisions. The company partners with leading hospitals and life sciences organizations to transform care delivery for cardiovascular patients. The company is based in Aliso Viejo, Calif., for more information, visit www.egnitehealth.com.